Ceratitis capitata (Wiedemann)
Taxonomic placing: Insecta, Holometabola, Diptera, Brachycera, Tephritidae.
Common name: Medfly, Mediterranean fruit fly.
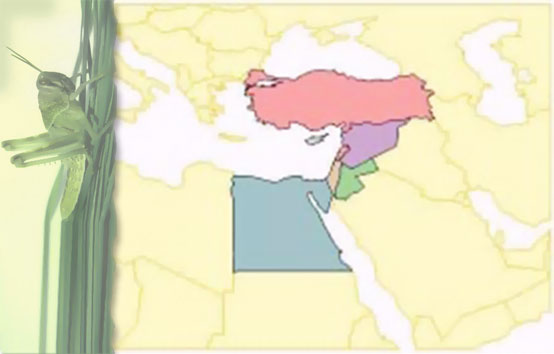
Geographical distribution: Africa, Mediterranean region, Southern Europe, Indian sub-continent, Western Australia, California, Hawaii.
Host plants: Several hundred deciduous, subtropical, and tropical fruit trees and some vegetables.
Morphology: The maggot is whitish-yellow, about 7-9 mm in length, elongate, its anterior end narrowed, caudal segments broader, with a pair of posterior spiracles. The adult fly is 3.5 to 5 mm in length, with red eyes, dorsum dark with characteristic pale markings, venter yellow, wings hyaline with yellow to dark spots. The male bears a pair of dark spatula-like setae on its head.
Life cycle: Females lay their eggs (about 300 and sometimes more) under the skins of ripening fruits, preferably where the skin is soft or wounded. The emerging larvae develop inside the fruit, dropping off to pupate in the soil. A total life cycle may take several weeks, depending on the host-fruit: a fortnight in grapefruits, almost 4 weeks in loquat, both at 24ºC. The adults, which can live for several months, do not disperse much. Under mild temperature conditions the pest can raise 3-4 annual generations, whereas in warmer regions it may have 6-8 generations; the medfly has no diapause. As the threshold for development is around 16ºC, in most Middle East regions the medfly can reproduce the year around, different available fruits providing oviposition sites.
Economic importance: The medfly is among the world’s most destructive fruit pests. The maggots tunnel in fruits and feed on the pulp, eventually reducing the whole to a juicy, inedible mass. Citrus fruit, especially oranges, grapefruit and mandarins are heavily attacked and damaged, as are apples, figs, pears and many subtropical fruits. Some countries enforce strict quarantine measures against this pest, which had spread throughout the world due to the global fruit trade. The medfly’s economic injury level is very low, and the presence of even a single fly could trigger control measures.
Management
Monitoring: Fruit sampling was formerly a common method of monitoring medfly populations. Another method is using yellow sticky-board traps that attract the medfly due to their color and to which the insects stick and can then be counted. Methods in more recent use are ammonia traps to bait females and attractants (like trimedlure) for males. An example is the widely used McPhail trap. It consists of a bell-shaped receptacle open at the bottom containing an attractant along with a toxicant, that may continue to attract for 2-3 months. Other attractants and traps (e.g. Jackson trap, Steiner trap, Multilure trap and others) have also been developed. The effectiveness of control measures is assessed by the punctured and dropped fruits and by the number of males captured in traps.
Horticultural methods: Heavy, continuous infestations may be contained by removing all fruit from potential host trees, and burying them within plastic bags. McPhail traps are also used for mass-trapping, as a “lure-and-kill” technique. Its efficacy depends on the number of traps placed in an orchard, on their location (e.g. on its borders to catch invading flies), on the crop to be defended, and on the pest’s population density. Some lures also attract parasitoids, but spinosad or the insecticidal paint phloxin B do not harm these natural enemies.
Chemical control: Two main pesticidal methods were used in different parts of the world to control the medfly. One consists of drenching with a pesticide the soil under host trees, where the pest is known to pupate. Another is spraying the trees with a suitable insecticide, the organophosphate malathion in the past, spinosad more recently. As noted, the baits also kill the flies due to included pesticides. Treatments with Kaolin dust provided better medfly control than spinosad or malathion in Tunisia, although kaolin may affect natural enemies.
Chemosterilization: Male and female medflies are attracted to a device that dispenses ammonia and trimedlure and contains an insect growth regulator. This reduces the viability of eggs laid by the exposed females as well as by wild females that had mated with exposed males.
The biofeed method: This consists of attracting the pests to a yellow suspended device with a sticky bait that contains spinosad. The flies feed on it, cannot escape and die. This device remains active for several months and is suitable for both conventional and organic farmers.
Sterile medfly release: This consists of mass-rearing the flies, killing the females by heat and sterilizing the males by irradiation. These males are then released in very large numbers (about 80-100 sterilized males to a single wild fly, the appropriate ratios being obtained from prior monitoring). Wild medfly females that mate with sterile males produce no offspring and due to the great preponderance of the sterile males, the pest population dies out. Medfly control in large areas is achieved by releasing the sterilized males from airplanes, operations that often requires international cooperation, as is happening in the Middle East
Genetic manipulation: A transgenic medfly strain, whose males carry a dominant lethal gene, was developed by molecular methods. When such males mate with wild females they induce embryonic lethality in the resulting eggs, which do not hatch.
Biological control: Several ant species and predatory Coleoptera prey on medfly stages in the soil. In different parts of the world the larvae are attacked by various hymenopterous parasitoids. These include Aganaspis daci and Psyttalia concolor, Diachasmimorpha kraussii, Diachasmimorpha tryoni (Cameron) and Fopius ceratitivorus. A drawback of this approach is that the parasitoids are unable to control medfly infestations to below the extremely low economic threshold.
References
Alemany. A., Gonzales. A., Juan, A. and Tur, C. 2008. Evaluation of a chemosterilization strategy against Ceratitis capitata (Diptera: Tephritidae) in Mallorca island (Spain). Journal of Applied Entomology 132: 746-52.
Argov, Y. and Gazit Y. 2008. Biological control of the Mediterranean fruit fly in Israel: introduction and establishment of natural enemies. Biological Control 46: 502-7.
Braham, M., Pasqualini, E. and Ncira, N. 2007. Efficacy of kaolin, spinosad and malathion against Ceratitis capitata in Citrus orchards. Bulletin of Insectology 60: 39-47.
Delrio, G, and Cocco, A. 2011. Tephritidae. In Vacante V. and Gerson, U. (eds). Integrated Cotrol of Citrus Pests in the Mediterranean Region. Bentham E-book, Science Publishers, pp. 206-222.
Epsky, N.D., Hendrichs, J. (and 8 co-authors) 1999. Field evaluation of female-targeted trapping systems for Ceratitis capitata (Diptera: Tephritidae) in seven countries. Journal of Economic Entomology 92: 156-164.
Israely, N. and Ben-Yehuda, S. 2006. Biofeed: an environmentally friendly control device for the Mediterranean fruit fly.
Phytoparasitica 34: 383-384.
Schetelig, M.F., Caceres, C., Zacharopoulou, A., Franz, G. and Wimmer, E.A. 2009. Conditional embryonic lethality to improve the sterile insect technique in Ceratitis capitata (Diptera: Tephritidae). BMC Biology 7(4):10.1186⁄1741-7007/7/4.
Vargas, R. I., Miller, N. W. and Prokopy R. J. 2002. Attraction and feeding responses of Mediterranean fruit fly and a natural enemy to protein baits with two novel toxins, phloxine B and spinosad. Entomologia Experimentalis et Applicata 102: 273-282.
Urbaneja, A., Garcìa Marì, F. (and 5 co-authors). 2006. Influence of ground predators on the survival of the Mediterranean fruit fly pupae, Ceratitis capitata, in Spanish citrus orchards. BioControl 51: 611-26.
http://entnemdept.ifas.ufl.edu/creatures/fruit/mediterranean_fruit_fly.htm
http://www.plants.org.il/uploadimages/The%20Mediterranean%20Fruit%20Fly%20-%20English%20version.pdf